|
This makes it easier to understand and predict how atoms will interact to form chemical bonds. The cobalt orbital diagram is a graphical representation of the electron configuration of the cobalt atom. The configuration notation provides an easy way for scientists to write and communicate how electrons are arranged around the nucleus of an atom. This give us the (correct) configuration of:įor the Cu+ ion we remove one electron from 4s1 leaving us with:įor the Cu2+ ion we remove a total of two electrons (one from the 4s1 and one form the 3d10) leaving us with Therefore, one of the 4s2 electrons jumps to the 3d9. Half-filled and fully filled subshell have got extra stability. In a D4h site with a strong elongation the 3A2g term of cobalt (+III) can be stabilized with an electronic configuration intermediate between low spin. What is the electron configuration of Co + Who invented Cobalt Where Why is Cobalt-60 used to treat cancer What are cobalt compounds used to make Cobalt reacts with what to produce hydrogen gas When is superalloys use preferred What are the common ores of cobalt Name one of the uses of cobalt-60, other than cancer treatment. As stated, you could simply count the boxes on the periodic table, and since Cobalt is the 7th element of the first row transition metals, we get Co: Ar 4s 2 3d 7. The reason why it is 3d 7 can be explained using the periodic table. Therefore we have (still incorrect) 1s 22s 22p 63s 23p 63d 94s 2Ĭorrect Electron Configuration for Copper (Cu) Thus, the electron configuration for Cobalt at ground state would simply be Co: Ar 4s 2 3d 7. Cobalt's electron configuration is 3d 7 4s 2 1s 2 2p 6 3s 2 3d 7 4s 2, 8, 14, 22. Both of the configurations have the correct numbers of electrons in each orbital, it is just a matter of how the electronic configuration notation is written ( here is an explanation why). The web page lists the electron configuration of all elements from hydrogen to cobalt, with shorthand and full forms. Note that when writing the electron configuration for an atom like Cu, the 3d is usually written before the 4s. Therefore the expected electron configuration for Copper will be 1s 22s 22p 63s 23p 64s 23d 9. After the 4s is full we put the remaining six electrons in the 3d orbital and end with 3d9. We now shift to the 4s orbital where we place the remaining two electrons. The La 2 Li 0.5 Co 0.5 O 4 phase, whose layer structure favours strongly such a distortion of the CoO 6 octahedra. Since the 3s if now full we'll move to the 3p where we'll place the next six electrons. In a D 4h site with a strong elongation the 3 A 2g term of cobalt (+III) can be stabilized with an electronic configuration intermediate between low spin ( 1 A 1g) and high spin ( 5 E g ): d 2xz d 2yz d 1xy d 1z 2d 0x 2 y 2. We'll put six in the 2p orbital and then put the next two electrons in the 3s. The p orbital can hold up to six electrons. The next six electrons will go in the 2p orbital. Since 1s can only hold two electrons the next 2 electrons for Copper go in the 2s orbital. In writing the electron configuration for Copper the first two electrons will go in the 1s orbital. Write the electron configuration and orbital diagram for Co (cobalt) in its ground state. The noble gas configuration for sodium is Ne 3s 1. So, the noble gas core symbol Ne replaces that portion of the sodium electron configuration. The electron configuration of neon is 1s 2 2s 2 2p 6. Using Noble Gas Notation you can write the s,p,d,f configuration in an abbreviated form.Video: Cu, Cu +, and Cu 2+ Electron Configuration Notation Looking at a periodic table, note the noble gas before sodium is neon. So the entire configuration is as follows:Īs another example, Argon is a noble gas and has each of the electron levels filled. The ground state electron configuration of ground state gaseous neutral cobalt is Ar.3d7. However, the inner transition metals or d block are always one level lower than the energy level that the element is found on meaning the electron configuration for cobalt must end as #3d^7#.Īll electron levels must be filled above that point. Cobalt atoms have 27 electrons and the shell structure is 2.8.15.2. 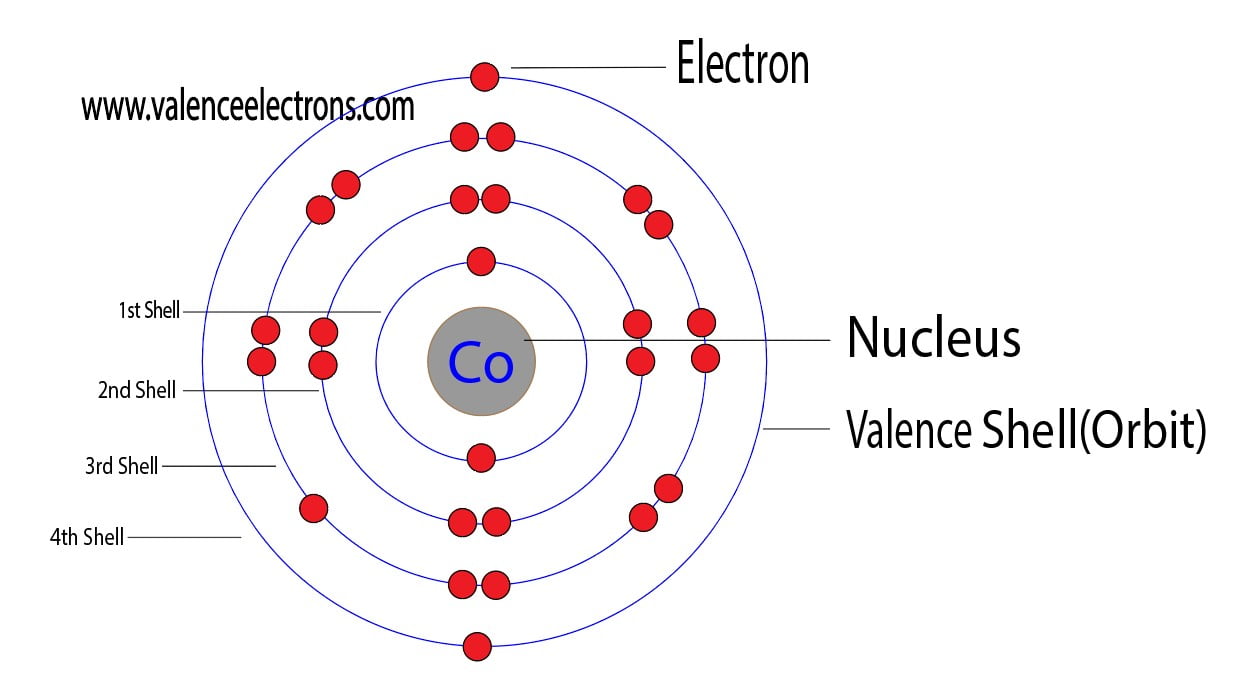
The ground state electron configuration of ground state gaseous neutral cobalt is Ar. The element cobalt can be found in the 4th row or 4th energy level of the periodic table. Electron Configuration Abbreviated Form Boron (5) 1s22s22p1 He2s22p1. Cobalt is in the 7th column of the d block and therefore has 7 d electrons #d^7#. The s,p,d,f configuration for cobalt (Co) is #1s^2 2s^2 2p^6 3s^2 3p^6 4s^2 3d^7#, determined by the position of the element on the periodic table.Ĭobalt is an inner transition metal which means the electron configuration will end in a d block.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
